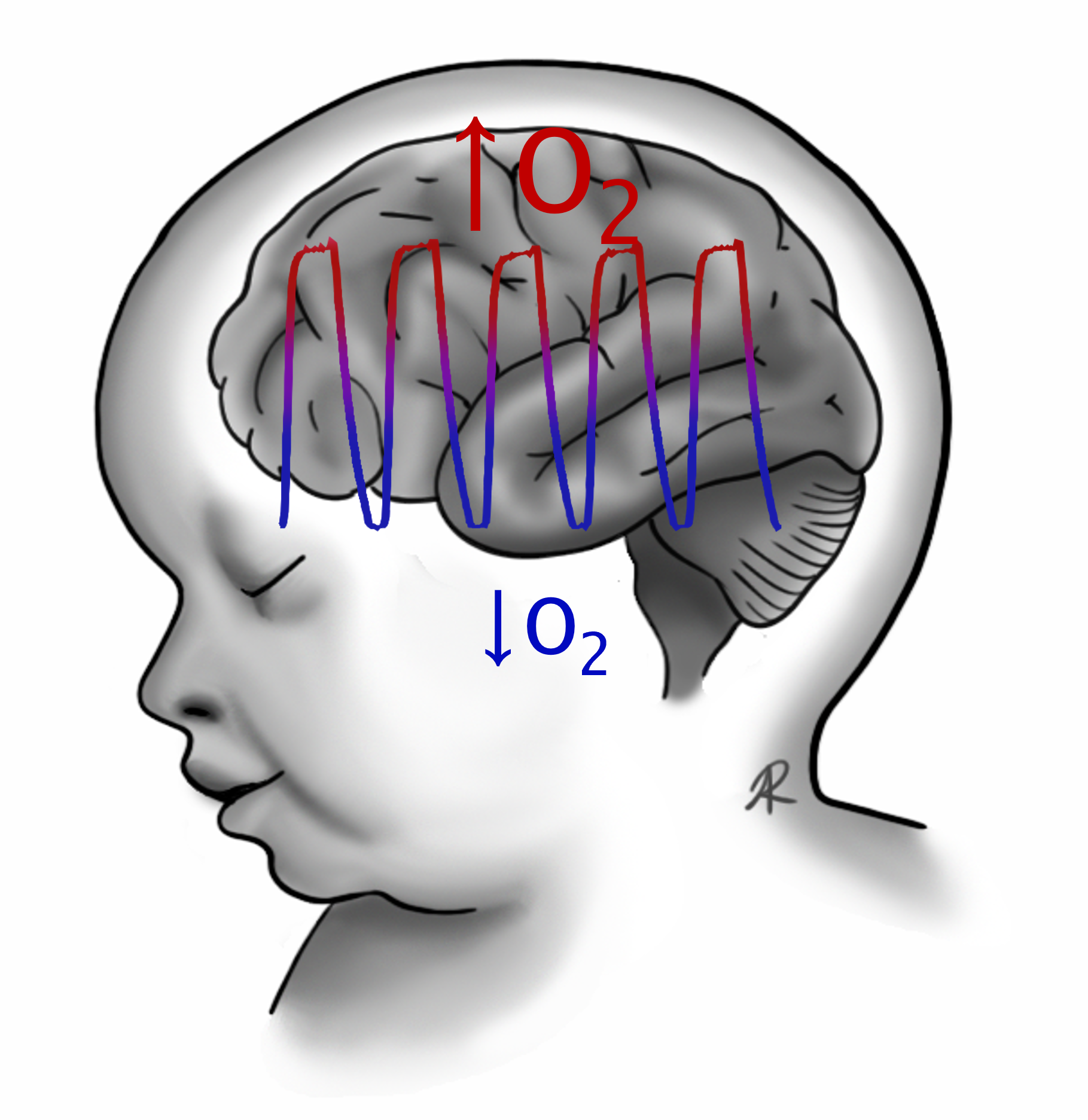

The cerebellum is involved in diverse functions, from motor control and equilibrium to spatial learning and emotion. At birth, the human cerebellar cortex is still immature, making it vulnerable to perinatal events such as hypoxia. Apnea of prematurity (AOP) is a main cause of perinatal hypoxia, as it occurs in over 50% of preterm infants. Several relationships between cerebellar functions and the deficits observed in children having suffered from AOP have been demonstrated, but the physio-pathological mechanisms by which AoP affects the cerebellum remain poorly understood.
This work aims at shedding light on the mechanisms underlying cerebellar hypoxic injury. To this end, we developed an intermittent hypoxia (IH) protocol, consisting of repeated 2-minute cycles of hypoxia-reoxygenation (including 20 seconds at 5% O2), applied between P2 and P12, 6 hours per day, which constitutes a valid murine model of AOP.
In a first study, we show that the cerebellum is indeed sensitive to IH, and presents a significant delay in maturation at the end of our IH protocol. In addition, hypoxia seems to induce an overexpression of genes involved in reactive oxygen species (ROS) production, which accumulate in the cerebellum. Conversely, genes coding for antioxidant enzymes are underexpressed after IH, suggesting a failure of the defense system against ROS. IH also induces long-term damage, in the form of learning and motor deficits, which are associated with the over-innervation of Purkinje cells by climbing fibers.
We followed these results with a transcriptomic study of genes involved in cell differentiation and migration. We analyzed the expression of these genes by RT-qPCR, in different developmental stages and in different cell types, using laser capture microdissection to separate cerebellar layers. This allowed us to determine that the period most vulnerable to IH is the P8 stage in mice, during which a significant number of genes are dysregulated in all cerebellar cortical layers. Moreover, it seems that all processes involved in cerebellar development are impacted, including proliferation, differentiation, synaptic connectivity, and myelination. Several compensatory and neuroprotective mechanisms are put in place during and after IH, but they do not seem to be sufficiently effective considering the persistence of deficits through adulthood.
Finally, given the established connection between hypoxia and vascularization, we tested a gene panel focused on these processes and found significant regulations of angiogenic and growth factors. In addition, preliminary imaging results suggest that IH cerebella have a looser and less voluminous vascular network, a factor that could participate in the pathophysiology of AOP. We aim to pursue this line of inquiry with additional immunohistochemical and clearing studies.
Overall, the data presented here demonstrate that the cerebellum is affected by IH, and that its injury is, at least in part, responsible for the symptoms observed in children having experienced AOP. This project provides elements to better understand AOP, by connecting behavioral and histological alterations to plausible underlying cellular and molecular mechanisms. In the long term, it could lead to the identification of novel therapeutic targets to improve the current clinical management of this highly prevalent pathology.